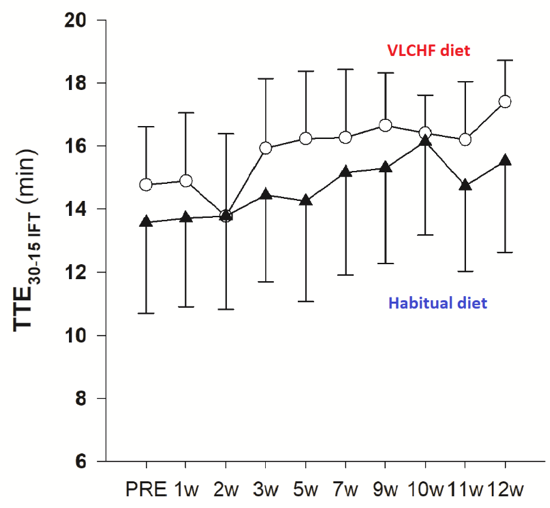
Figure 1. Total time to exhaustion in 30-15 Intermittent Fitness Test (TTE30-15IFT )
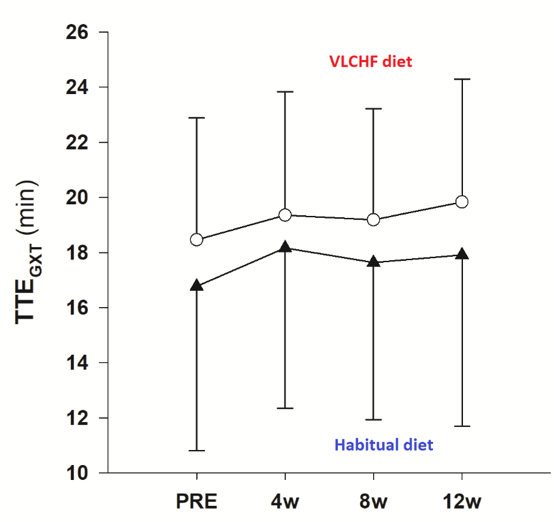
Figure 2. Total time to exhaustion in the Graded Exercise Test to volitional exhaustion (TTEGXT)
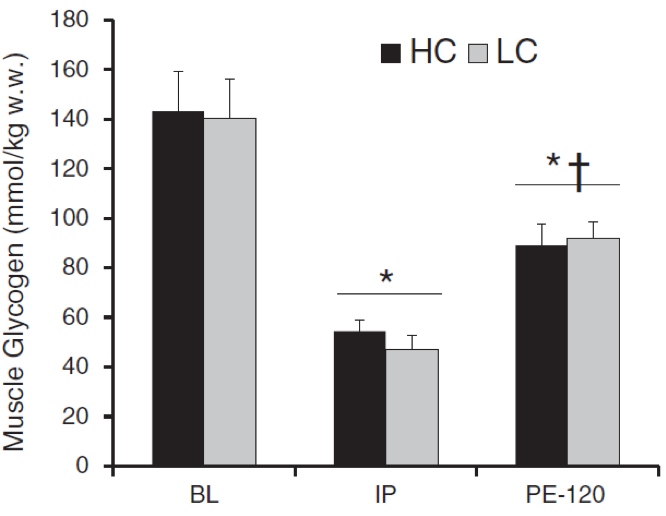
Figure 3. Muscle Glycogen changes after a treadmill run at 64 % VO2max for 180 min from the study of Volek et al. (23).
Legend: HC/LC – high/low carbohydrate, BL – baseline (pre-exercise), IP – immediate post-exercise, PE-120 – 120 min post-exercise. * significant difference from baseline, ⴕ – a significant difference from IP.
 About the author: Lukas Cipryan is an associate professor at the University of Ostrava (Czechia) specializing in exercise physiology, sport training and nutrition. As a former ice hockey player, he spent nearly 10 years coaching youth ice hockey players while being embedded within professional ice hockey and volleyball teams as a strength & conditioning coach. Currently, in addition to his academic role, he works for the football academy of FC Banik Ostrava. Follow him on Twitter @LukasCipryan.
References:
About the author: Lukas Cipryan is an associate professor at the University of Ostrava (Czechia) specializing in exercise physiology, sport training and nutrition. As a former ice hockey player, he spent nearly 10 years coaching youth ice hockey players while being embedded within professional ice hockey and volleyball teams as a strength & conditioning coach. Currently, in addition to his academic role, he works for the football academy of FC Banik Ostrava. Follow him on Twitter @LukasCipryan.
References:
- Branco, AF, Ferreira, A, Simões, RF, Magalhães-Novais, S, Zehowski, C, Cope, E, et al. Ketogenic diets: from cancer to mitochondrial diseases and beyond. Eur J Clin Invest 46: 285–298, 2016.Available from: http://doi.wiley.com/10.1111/eci.12591
- Broom, GM, Shaw, IC, and Rucklidge, JJ. The ketogenic diet as a potential treatment and prevention strategy for Alzheimer’s disease. Nutrition 60: 118–121, 2019.Available from: https://doi.org/10.1016/j.nut.2018.10.003
- Burke, LM and Hawley, JA. Effects of short-term fat adaptation on metabolism and performance of prolonged exercise. Med Sci Sports Exerc 34: 1492–8, 2002.Available from: http://www.ncbi.nlm.nih.gov/pubmed/12218744
- Burke, LM and Hawley, JA. Swifter, higher, stronger: What’s on the menu? Science 362: 781–787, 2018.Available from: http://www.ncbi.nlm.nih.gov/pubmed/30442803
- Burke, LM, Ross, ML, Garvican-Lewis, LA, Welvaert, M, Heikura, IA, Forbes, SG, et al. Low carbohydrate, high fat diet impairs exercise economy and negates the performance benefit from intensified training in elite race walkers. J Physiol 595: 2785–2807, 2017.Available from: http://doi.wiley.com/10.1113/JP273230
- Cahill, GF. Fuel Metabolism in Starvation. Annu Rev Nutr 26: 1–22, 2006.Available from: http://www.annualreviews.org/doi/10.1146/annurev.nutr.26.061505.111258
- Cipryan, L, Plews, DJ, Ferretti, A, Maffetone, PB, and Laursen, PB. Effects of a 4-Week Very Low-Carbohydrate Diet on High-Intensity Interval Training Responses. J Sports Sci Med 17: 259–268, 2018.Available from: http://www.ncbi.nlm.nih.gov/pubmed/29769827
- Dostal, T, Plews, DJ, Hofmann, P, Laursen, PB, and Cipryan, L. Effects of a 12-week very-low carbohydrate high-fat diet on maximal aerobic capacity, high-intensity intermittent exercise, and cardiac autonomic regulation: non-randomized parallel-group study. Front Physiol , 2019.
- Feinman, RD, Pogozelski, WK, Astrup, A, Bernstein, RK, Fine, EJ, Westman, EC, et al. Dietary carbohydrate restriction as the first approach in diabetes management: Critical review and evidence base. Nutrition 31: 1–13, 2015.Available from: http://dx.doi.org/10.1016/j.nut.2014.06.011
- Hansen, NW, Hansen, AJ, and Sams, A. The endothelial border to health: Mechanistic evidence of the hyperglycemic culprit of inflammatory disease acceleration. IUBMB Life 69: 148–161, 2017.Available from: http://doi.wiley.com/10.1002/iub.1610
- Kwon, Y-J, Lee, H-S, and Lee, J-W. Association of carbohydrate and fat intake with metabolic syndrome. Clin Nutr 6–11, 2017.Available from: http://linkinghub.elsevier.com/retrieve/pii/S0261561417302339
- Langfort, J, Zarzeczny, R, Pilis, W, Nazar, K, and Kaciuba-Uscitko, H. The effect of a low-carbohydrate diet on performance, hormonal and metabolic responses to a 30-s bout of supramaximal exercise. Eur J Appl Physiol 76: 128–133, 1997.Available from: http://link.springer.com/10.1007/s004210050224
- Lima-Silva, AE, Pires, FO, Bertuzzi, R, Silva-Cavalcante, MD, Oliveira, RSF, Kiss, MA, et al. Effects of a low- or a high-carbohydrate diet on performance, energy system contribution, and metabolic responses during supramaximal exercise. Appl Physiol Nutr Metab 38: 928–934, 2013.Available from: http://www.nrcresearchpress.com/doi/abs/10.1139/apnm-2012-0467
- Ludwig, DS, Willett, WC, Volek, JS, and Neuhouser, ML. Dietary fat : From foe to friend ? 770: 764–770, 2018.
- Maffetone, PB and Laursen, PB. Athletes: Fit but Unhealthy? Sport Med – Open 2: 24, 2016.Available from: http://www.ncbi.nlm.nih.gov/pubmed/27340616%5Cnhttp://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=PMC4882373
- Nieuwdorp, M, van Haeften, TW, Gouverneur, MCLG, Mooij, HL, van Lieshout, MHP, Levi, M, et al. Loss of endothelial glycocalyx during acute hyperglycemia coincides with endothelial dysfunction and coagulation activation in vivo. Diabetes 55: 480–6, 2006.Available from: http://www.ncbi.nlm.nih.gov/pubmed/16443784
- Noakes, TD and Windt, J. Evidence that supports the prescription of low-carbohydrate high-fat diets: a narrative review. Br J Sports Med 51: 133–139, 2017.Available from: http://bjsm.bmj.com/lookup/doi/10.1136/bjsports-2016-096491
- O’Keeffe, KA, Keith, RE, Wilson, GD, and Blessing, DL. Dietary carbohydrate intake and endurance exercise performance of trained female cyclists. Nutr Res 9: 819–830, 1989.Available from: http://linkinghub.elsevier.com/retrieve/pii/S0271531789800272
- Pitsiladis, YP and Maughan, RJ. The effects of exercise and diet manipulation on the capacity to perform prolonged exercise in the heat and in the cold in trained humans. J Physiol 517: 919–930, 1999.Available from: http://doi.wiley.com/10.1111/j.1469-7793.1999.0919s.x
- Schmidt, S, Christensen, MB, Serifovski, N, Damm‐Frydenberg, C, Jensen, J-EB, Fløyel, T, et al. Low versus High Carbohydrate Diet in Type 1 Diabetes: A 12‐week randomized open‐label crossover study. Diabetes, Obes Metab dom.13725, 2019.Available from: https://onlinelibrary.wiley.com/doi/abs/10.1111/dom.13725
- Starling, RD, Trappe, TA, Parcell, AC, Kerr, CG, Fink, WJ, and Costill, DL. Effects of diet on muscle triglyceride and endurance performance. J Appl Physiol 82: 1185–1189, 1997.
- Thomas, DT, Erdman, KA, and Burke, LM. Position of the Academy of Nutrition and Dietetics, Dietitians of Canada, and the American College of Sports Medicine: Nutrition and Athletic Performance. J Acad Nutr Diet 116: 501–528, 2016.
- Volek, JS, Freidenreich, DJ, Saenz, C, Kunces, LJ, Creighton, BC, Bartley, JM, et al. Metabolic characteristics of keto-adapted ultra-endurance runners. Metabolism 65: 100–110, 2016.Available from: http://linkinghub.elsevier.com/retrieve/pii/S0026049515003340
- Volek, JS, Noakes, T, and Phinney, SD. Rethinking fat as a fuel for endurance exercise. Eur J Sport Sci 15: 13–20, 2015.Available from: http://www.ncbi.nlm.nih.gov/pubmed/25275931






